|
Thomsons, which was the first model of Atom. Learn more about Thomsons Model of an Atom, by J.J. The size of the nucleus of an atom is very small in comparison to the total size of an atom.A strong electrostatic force of attractions holds together the nucleus and electrons. An atom has no net charge or they are electrically neutral because electrons are negatively charged and the densely concentrated nucleus is positively charged. 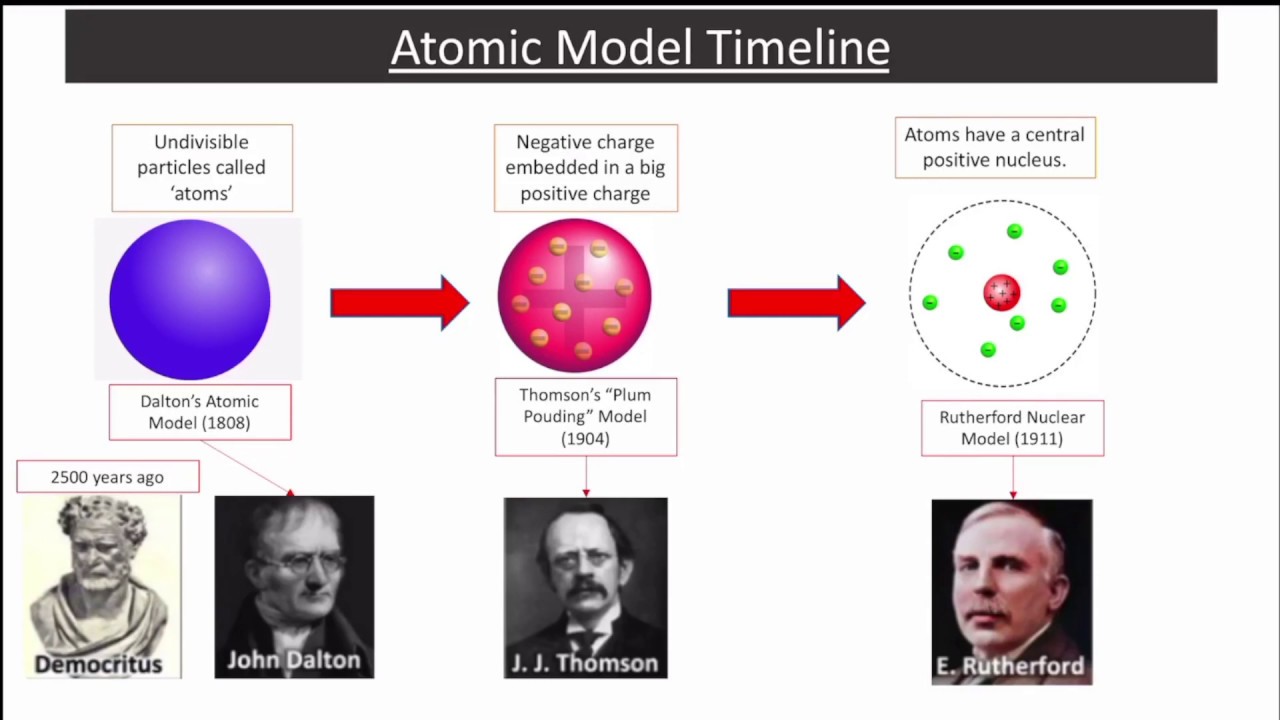
These fixed circular paths were termed as “orbits.” The electrons revolve around the nucleus in a fixed circular path at very high speed. 
Atoms nucleus is surrounded by negatively charged particles called electrons.It was found out later that the very small and dense nucleus of an atom is composed of neutrons and protons. This region of the atom was called as the nucleus of an atom. Majority of the mass of an atom was concentrated in a very small region. An atom is composed of positively charged particles.What is the difference between the Thomsons and Rutherford Atomic model? Postulates of Rutherford atomic model based on observations and conclusions Therefore, he concluded that the positively charged particles covered a small volume of an atom in comparison to the total volume of an atom. Moreover, very few particles had deflected at 180 o. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
